Photochemistry of light-responsive proteins
Light-responsive proteins have evolved to use light to drive complex processes ranging from
photosynthesis to vision. These systems not only serve as an inspiration for technologies but have
also been implemented directly in biotechnologies such as bioimaging, biosensing, optogenetics,
and photodynamic therapy. However, engineering photoreceptors for use in biotechnology requires
a fundamental understanding of how they operate on a molecular level. To this end, we use hybrid
quantum mechanical/molecular mechanical models to understand how biological systems respond
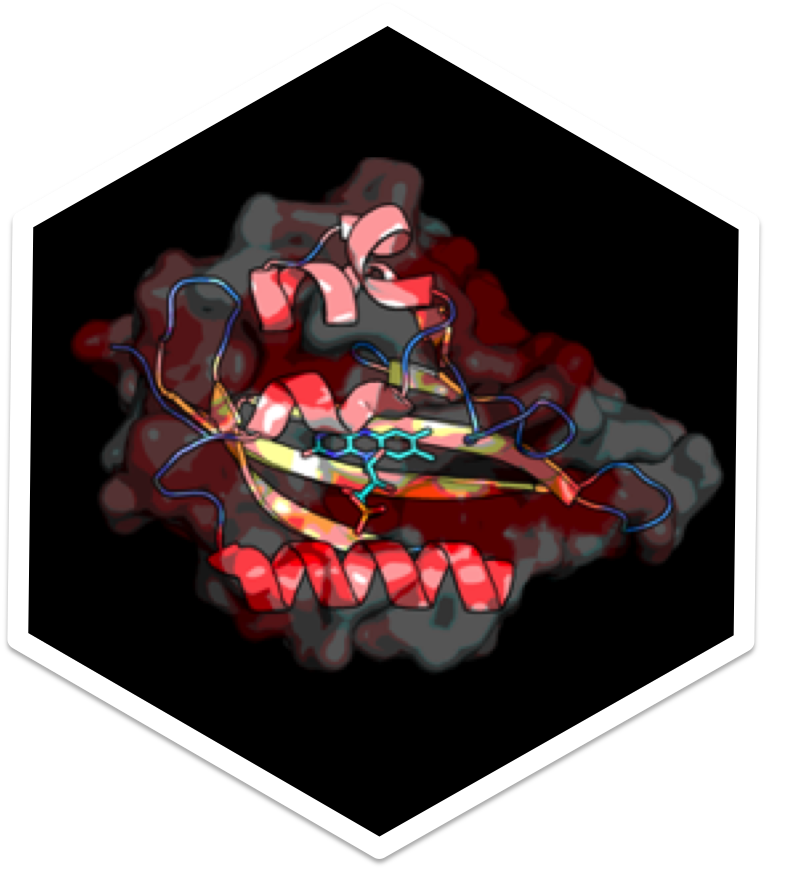
to light. We are currently focused on studying light-oxygen-voltage (LOV) domains.
Investigating new classes of fluorescent proteins
Flavin-binding fluorescent proteins (FbFPs) are recently engineered classes of
fluorescent proteins that have attractive properties. In particular, in comparison
to green fluorescent protein (GFP) derivatives, FbFPs are smaller (less genetic
content to express) and work in anaerobic conditions. We will employ hybrid
quantum mechanical /molecular mechanical (QM/MM) models to investigate
the spectral tuning mechanism and photophysics of these systems.
Condensed-phase computational spectroscopy
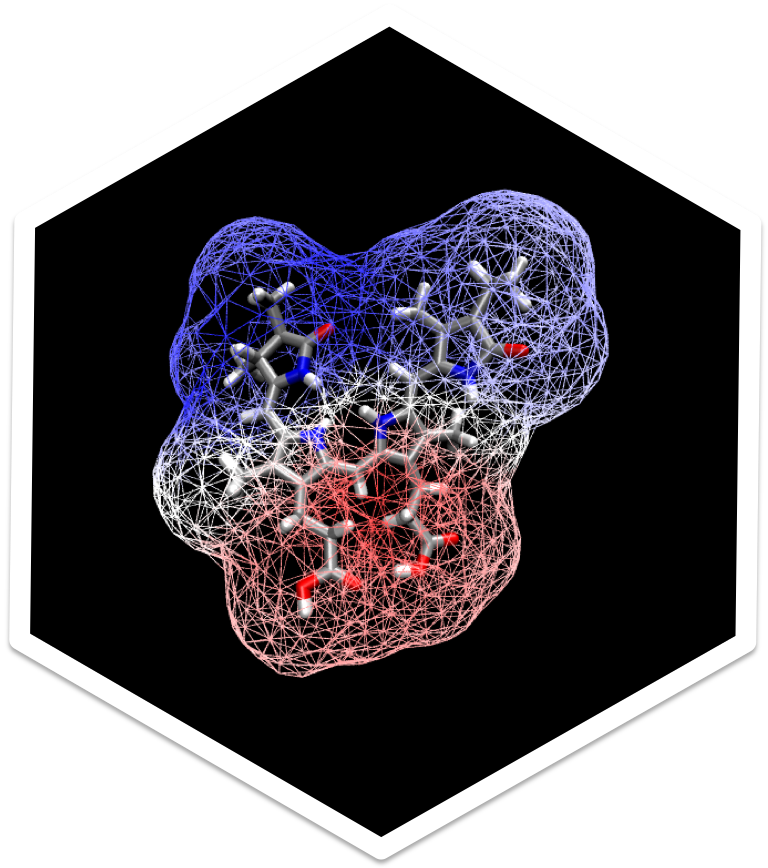
Spectroscopy is our window into the nanoscopic world of molecules. Gas-phase spectroscopies
probe the electronic, vibrational, and rotational structure and energetics of molecules with
remarkable precision. However, most of chemistry, and almost all of biology, happens in the
condensed phase, where spectra are shifted and broadened by interactions with biological
macromolecular structures and solvent molecules. These effects complicate the interpretation of
spectra, but they also provide essential information about the local molecular environment. We
study how solvent and protein interactions affect a molecule’s photoionization, absorption, and
emission spectra.
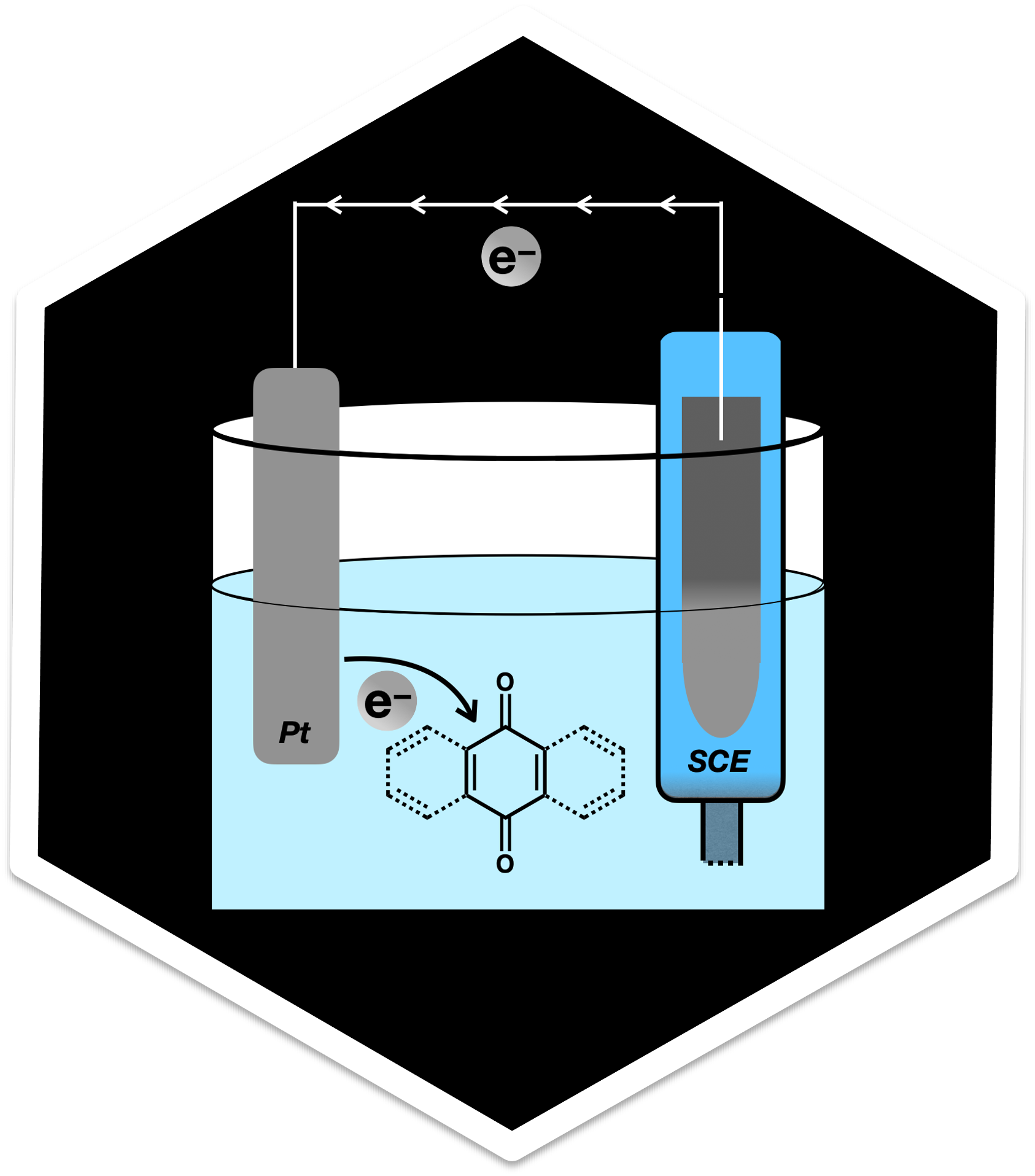
Redox and electron transfer chemistry
Reduction/oxidation (redox) reactions are ubiquitous in chemistry and nature.
Not only are many natural biosynthetic and regulatory processes based on
redox reactions, but redox biocatalysis also play a prominent role in chemical
industries. Our lab is interested in modeling redox potentials and electron
transfer processes in (photo)biocatalysts as well as in nanoscale materials
such as atomically precise nanoclusters.
Science Annex 504 and Natural Science Center 418
Department of Chemistry, Georgia State University, Atlanta, GA 30303.
Phone: (404)413-5569.
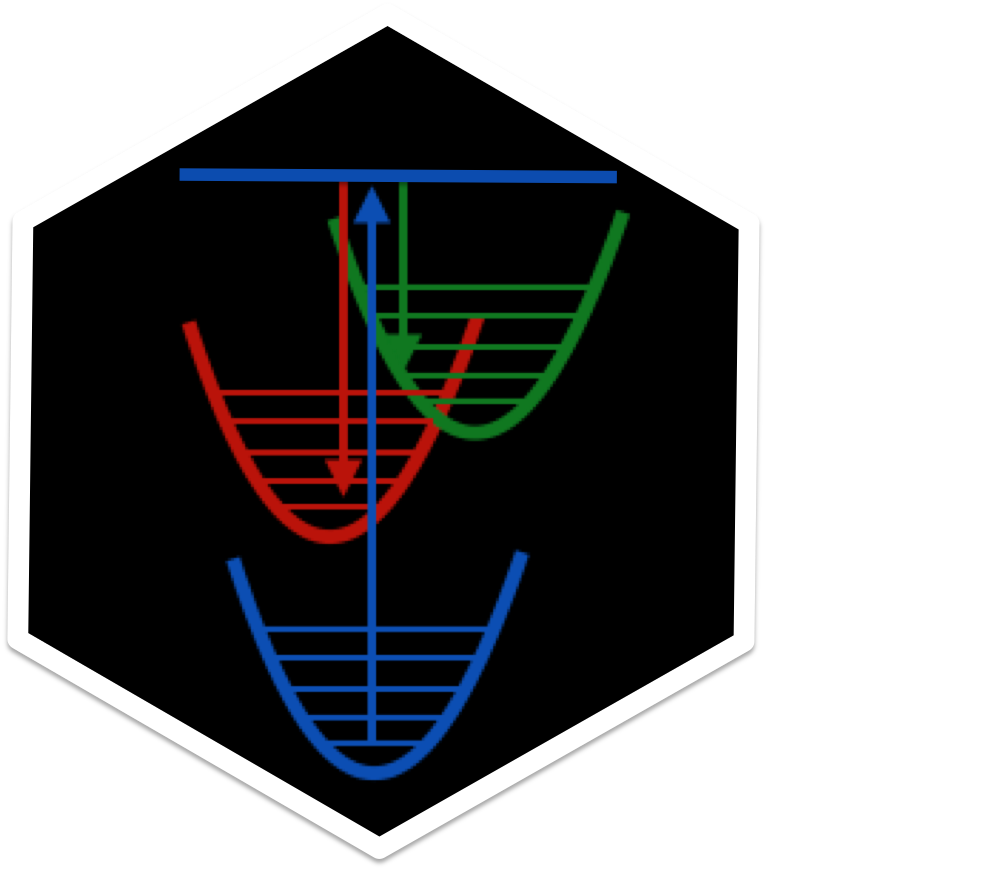
Other problems in photochemistry
We are broadly interested in modeling photophysical and photochemical
processes, and how they properties can be tuned by a solvent or
macromolecular (e.g., protein) environment.